About the Survey
ECRI Institute User Experience Network (UEN) surveys are designed to collect qualitative opinions from individuals and are not a statistical representation of a single organization or end-user population. UEN survey results are based solely on individual user responses at the time of the survey. Results do not reflect ECRI Institute opinions, recommendations, or endorsements of any kind. Because equipment configurations, clinical applications, and implementation strategies vary widely among facilities, participants’ impressions reflect their own unique experience and circumstances.
User experience data are independent of the clinical and scientific research and laboratory-based medical equipment evaluations conducted by ECRI Institute, and are simply one of several information sources to consider for technology evaluation, selection, and management. Other tools available to ECRI Institute members include: Results of formal device testing and evaluations conducted by ECRI engineers, clinicians, and research scientists; Technology reports published by ECRI that describe, review, or comment on device-related clinical literature; Device-related hazard reports and recalls published in ECRI Institute’s Alerts Tracker program; and pricing information available from ECRI Institute’s SELECTplus and PriceGuide programs. We strongly recommend that users consider all available sources of information and that no single source be utilized exclusively.
For more information about how our surveys are designed and distributed, as well as the methodology behind our analysis
read our User Experience Network FAQ.
Automated Medication Dispensing Cabinet User Experience Survey
Survey Conducted: May, 2015
User Experience Surveys were emailed to approximately 3,500 ECRI Institute member facilities. 159 survey responses were received and all respondents reported direct experience with one or more of the 18 Automated Medication Dispensing Cabinets listed on the survey form. Some users rated several different models. At least 1 survey response was received for 17 of the 18 models. The total number of responses received for all 18 models was 284. However, because 8 of the 18 models listed received less than 10 user responses, those models were excluded from our analysis.
The 8 Automated Medication Dispensing Cabinet Models excluded from our analysis because of small sample size (< 10 responses) are listed below:
(28 survey responses related to these 8 models were excluded from our analysis.)
- Aesynt – Anesthesia NarcStation
- Aesynt – Anesthesia Rx
- AmerisourceBergen – MedSelect Anesthesia
- AmerisourceBergen – MedSelect Flex
- AmerisourceBergen – MedSelect Flex with Centrak
- CareFusion – Pyxis Anesthesia ES
- Cerner - RxStation
- InterMetro - MedDispense
|
The remaining 256 survey responses related to the following 10 Medication Cabinets were analyzed by ECRI Institute:
-
Aesynt - AcuDose Rx
-
CareFusion - Pyxis Anesthesia 3500
-
CareFusion - Pyxis Anesthesia 4000
-
CareFusion - Pyxis CII Safe
-
CareFusion - Pyxis MedStation 3500
| -
CareFusion - Pyxis MedStation 4000
-
CareFusion - Pyxis MedStation ES
-
Omnicell - Anesthesia Workstation
-
Omnicell - OmniRx
-
Omnicell - OmniRx with OmniDispenser
|
Survey respondents were asked to rate the following items for each Model:

Scoring key:
-
1 = Very Poor
-
2 = Poor
-
3 = Average
-
4 = Good
-
5 = Very Good
Additional model-specific survey questions asked:

The following general questions were also asked:


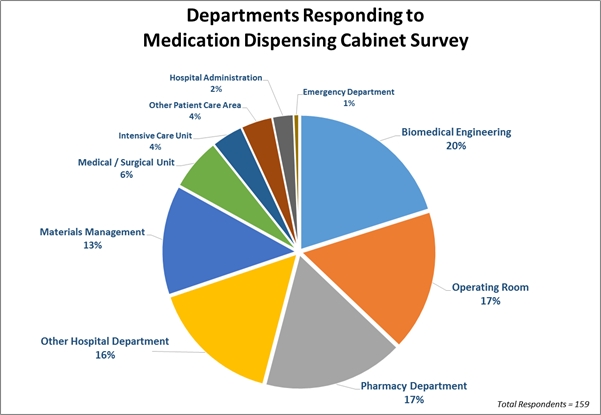
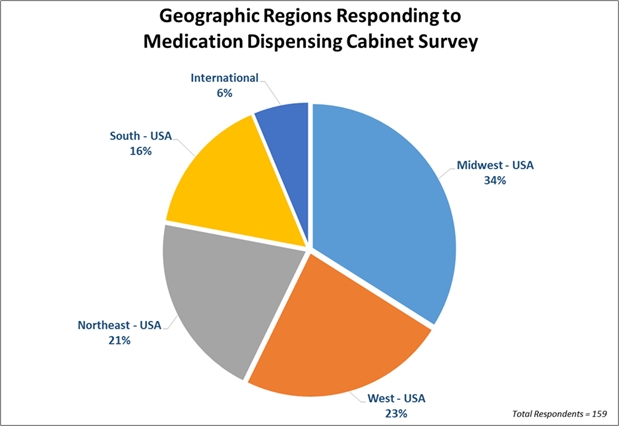
Survey Response Demographics



Policy Statement and Disclaimer
The information provided in this report may be derived from a number of sources, including manufacturers, clinical users, and member facilities. We make every reasonable effort to ensure that it is up-to-date and verify it with the most knowledgeable sources. This information has not been amplified, qualified, or confirmed by us through on-site observation, inspections, or review of hospital policies and procedures.
The contents of this document should not be regarded as a guarantee of the safety, performance, or cost-effectiveness of any equipment, device, or system. This analysis was created for ECRI Institute member organizations and is copyrighted. It may not be shared with manufacturers or any third party without written permission from ECRI Institute.
©2015 ECRI Institute. All Rights Reserved. This material is confidential, and use of the copyright notice is not intended to suggest or imply that the material has been published or is available for public dissemination. This report was created for ECRI Institute members and should not be reproduced or transmitted to third parties without written permission from ECRI Institute.