This Device Evaluation webcast is being made available to the general public. Contact us to learn about everything the Device Evaluation group can do for your organization.
This webcast was held on October 26, 2022. A recording of the event appears below.
Overview
Ventilator-associated pneumonia (VAP) is a serious and common complication for patients requiring multiple days of respiratory support. ECRI has reviewed the clinical evidence for a technology that may help reduce VAP incidence—endotracheal tubes fitted with subglottic secretion drainage ports—and is currently testing several devices in the lab. Watch a recording of our October 2022 webcast, which covered current strategies to reduce VAP, the evidence supporting use of subglottic drainage to prevent VAP, and ECRI's hands-on lab testing of subglottic drainage technology.
Presenters
- Jason Launders, Director of Operations, Device Evaluation, ECRI
- Ericka Kalp, PhD, MPH, CIC, FAPIC, Director of Consulting, PSRQ Risk Management Contracts, ECRI
- Savvas Pavlides, PhD, Senior Manager, Scientific Quality, Clinical Evidence-Genetic Test Assessment, ECRI
- Kathy Punzo, Senior Project Officer, Device Evaluation, ECRI
- Ismael Cordero, Senior Project Officer, Device Evaluation, ECRI
View a recording from the October 26, 2022, live-streamed lab webcast, "Exploring the Evidence and Devices Designed to Prevent Ventilator-Associated Pneumonia." Length: 0:00.
Play
The Evidence

|
Key Takeaways
 
|
Webcast Poll Results
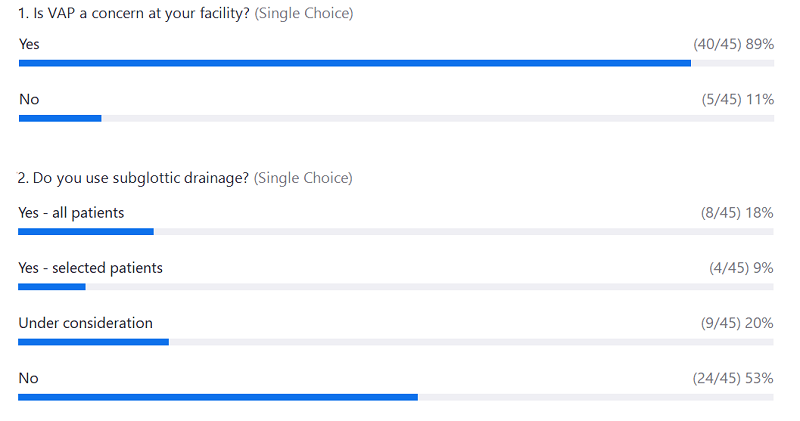
|
ECRI Resources
Clinical Evidence Assessment
Endotracheal Tubes with Subglottic Secretion Drainage Ports for Preventing Ventilator-associated Pneumonia
Guidance Articles
Medical Suction Devices: Using Them Safely and Effectively
Ventilator Safety and Ethics